Research Projects
We study the genetic regulation of growth and connectivity in the developing brain, and how cognition is shaped by the interplay of these “hard-wiring” processes and adaptation via plasticity. Thus, we investigate mechanisms of neurogenesis, gliogenesis, axon/dendrite development and synaptogenesis (i.e. growth and connectivity), as well as neuronal plasticity.
Our approach involves investigating genes associated with clinical conditions that feature abnormal development of the cerebral cortex, which is the largest structure in the brain and is responsible for our most sophisticated cognitive functions, including consciousness. The basic organization of the cerebral cortex is highly similar between humans and our model system of choice. This approach allows us to discover fundamental mechanisms by which the brain is built and then translate this into improved diagnostics and therapeutics for those affected by such conditions.
Autism encompasses a heterogeneous group of disorders that together make up autism spectrum disorder (ASD). ASD is defined by deficits in social interaction and communication, as well as repetitive behavior and restricted interests. ASD is highly heritable, indicating a strong genetic influence. Our lab is investigating the poorly understood problem of how genetic risk factors for ASD interact with genetic background and environmental factors to shape the spectrum of autism symptoms. We have discovered that autism-relevant symptoms can be modified by a “multiple-hit” mechanism that involves an interaction between a rare genetic variant that influences brain growth and structural connectivity (germline heterozygous PTEN mutation) and a common variant that influences functional connectivity and plasticity (variation in the serotonin transporter gene) in a preclinical model.
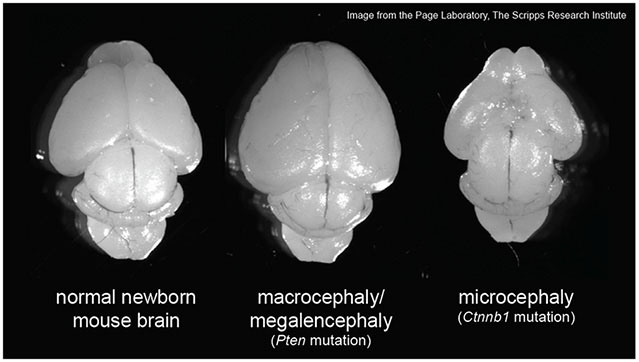
Macrocephaly/megalencephaly and microcephaly are characterized by extreme overgrowth or undergrowth of the brain, respectively. Macrocephaly and microcephaly are present in a subset of individuals with autism, intellectual disability and other neurodevelopmental disorders, and may represent a way to stratify patients into meaningful subgroups. Our work investigates genes associated with macrocephaly and microcephaly to elucidate molecular networks regulating brain growth and the development of behavior and cognition. We have discovered that macrocephaly and microcephaly genes interact to influence brain growth in a preclinical model, indicating that common biological pathways and cellular mechanisms might underlie these two conditions.
Our lab uses a variety of techniques, including transgenic and knockout models; genetics and genomics; developmental biology; neuroanatomy and histology; molecular cell biology; bioinformatics; pharmacology; imaging; and behavioral phenotyping.
Below are clips from an assay that we use to study social approach behavior in preclinical models that have undergone genetic or environmental manipulations relevant to autism. The top clip is of a wild type or “normal ” model, and the bottom clip is of a model that carries a mutation in Pten, a candidate susceptibility gene for autism. The preference of the model for interacting with a stimulus model (located in left chamber) is quantified during this assay.
Below are examples of preclinical models that we use to investigate the genetic basis of macrocephaly/megalencephaly and microcephaly.