Current Research at the Reeves Lab
Altered hyaluronan deposition by bronchial epithelial cells contributes to inflammation during respiratory syncytial virus infection
Lower respiratory tract infections commonly caused by respiratory syncytial virus (RSV) are a leading cause of hospitalizations and mortality worldwide in young children. Despite many years of study, the mainstay of treatment remains supportive care and currently no RSV specific treatment or vaccine is available.
The cells that line the airways, otherwise known as bronchial epithelial cells (BECs), are the primary target for RSV infection. Infection of the BECs with RSV leads to subsequent BEC damage, obstruction of the lower airways with cellular debris and mucous, and the establishment of airway inflammation by recruitment of immune cells such as neutrophils into the peribronchial space. Recent studies from our laboratory have demonstrated that RSV infection of stromal cells such as lung fibroblasts leads to the establishment of an extracellular matrix (ECM) that is enriched with hyaluronan (HA) which promotes the accumulation and activation of leukocytes in ex vivo cell culture models.
Furthermore, we found that the increased HA accumulation following RSV infection was more closely associated with the fibroblast cell layer and displayed greater modification with heavy chains (HC) which has been described in other studies to enhance the ECM’s ability to become sticky for inflammatory cells and promotes the inflammatory response in the lung.
The formation of HC-HA is the result of the enzymatic activity of tumor necrosis factor stimulated gene 6 (TSG-6), which is not normally expressed in healthy lung tissue, but is induced in response to injury. In our previous work with lung fibroblasts, we have shown that TSG-6 is upregulated during RSV infection and blocking its induction with siRNA decreased the amount of HC-HA that was formed and decreased the accumulation of leukocytes.
Despite being the primary target of RSV infection, no studies exist that have evaluated the effects of RSV infection on BEC production of HA or TSG-6 induced HC-HA formation despite the important role that HC-HA plays in promoting lung inflammation.
Our laboratory both has expertise in the characterization of HA matrices and access to primary human BECs from well-characterized pediatric donors placing our group in a unique position to evaluate the contribution of BEC derived HC-HA to the inflammatory response following RSV infection.
We hypothesize that RSV infection of BECs leads to increased production and accumulation of HA which will in turn promote the accumulation and activation of neutrophils in an ex vivo human cell culture model system.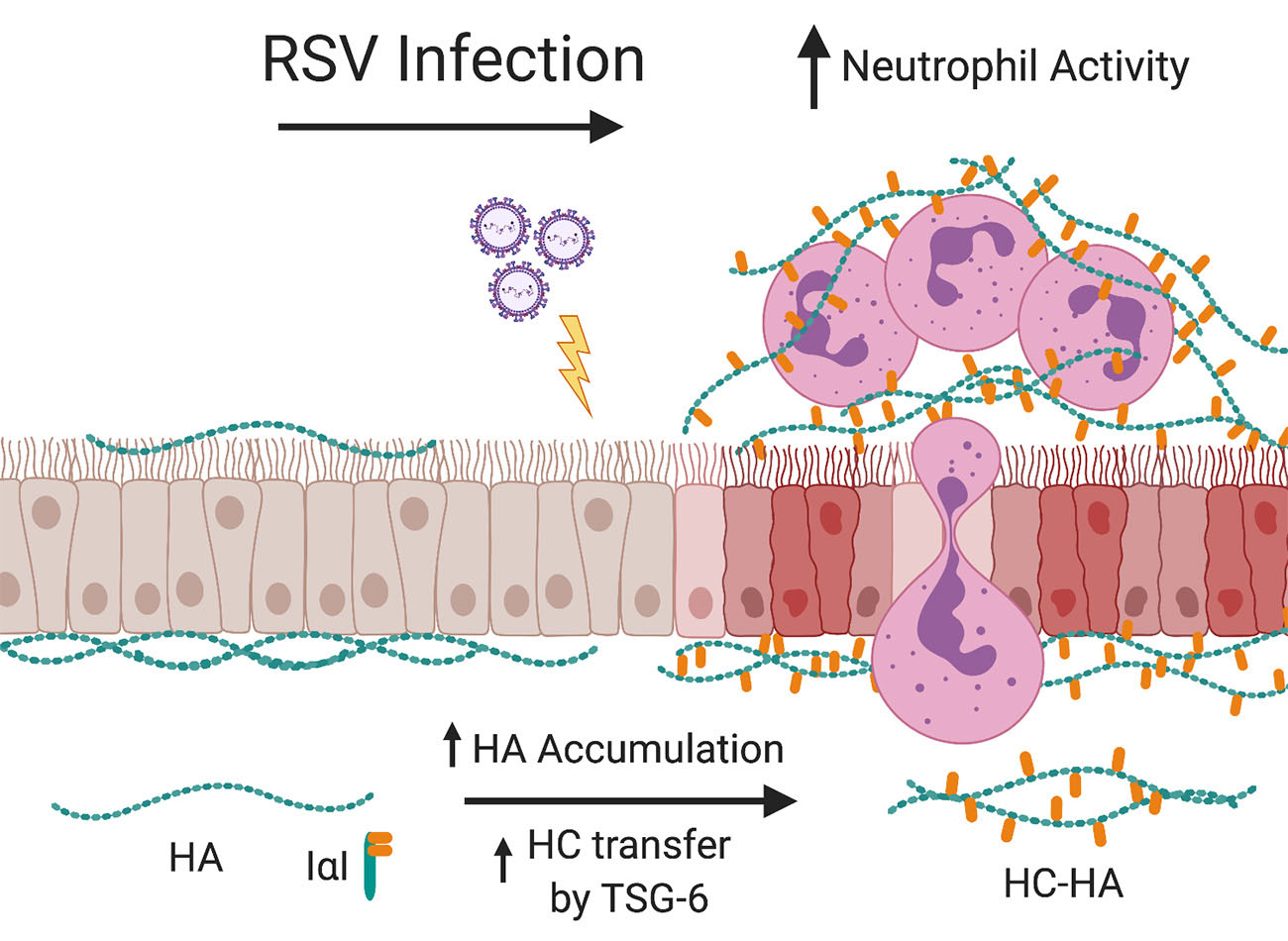
Additionally, we will test the hypothesis that RSV infection of BECs will induce the expression of TSG-6 thereby promoting the formation of HC-HA and further drive the accumulation and activation of neutrophils in our model system.
We will pharmacologically block the formation of HC-HA enriched ECMs and also block the induction of TSG-6 during RSV infection in order to establish whether the increased accumulation of the neutrophils is HC-HA dependent.
If true, characterizing the importance of HC-HA produced by BECs may lead to novel targets for therapeutic intervention during acute RSV infections and may be applicable to other respiratory viruses.
Developing an ex-vivo organotypic co-culture system to model airway epithelial cell-chondrocyte interactions during tracheal development
In this project, we study the effects of airway epithelial cell (AEC)-chondrocyte co-culture on hyaline cartilage formation and ECM deposition as a model of tracheal development.
Our group has an active research interest in the role of AECs in the coordination of airway remodeling and airway inflammation associated with alteration in the ECM during lung disease. Our lab has previously developed an ex vivo AEC-human lung fibroblast (HLF) co-culture model system to investigate the role of AEC-HLF crosstalk on ECM deposition and composition. In this project, we capitalize on the existing expertise with AEC-HLF co-culture model system to evaluate ECM changes to establish a new model of AEC-chondrocyte crosstalk to evaluate alterations in hyaline cartilage formation and ECM composition.
Using primary mesenchymal stromal cells (MSCs) differentiated into chondrocytes in ex vivo organoids, we will determine the effect of AEC-chondrocyte co-culture on the formation of hyaline cartilage and ECM deposition by co-cultured chondrocytes.
A primary goal of ours is to differentiate chondrocytes from MSCs obtained from pediatric donors with known FGFR2 craniosynostosis syndromes to be utilized in the AEC-chondrocyte co-cultures in collaboration with the Eldredge Lab. This will allow us to determine the effect of FGFR2 gain of function signaling on AEC differentiation and ciliogenesis compared to co-culture with chondrocytes differentiated from MSCs obtained from healthy donors.
We will also characterize the differences in the hyaline cartilage and ECM deposition by chondrocytes with a FGFR2 gain of function mutation co-cultured with AECs compared to co-cultured chondrocytes derived from unaffected donors.
These studies will give us insight to into the altered regulation of tracheal cartilage deposition that may underline the development of TCS.
