Stuart Lab
Research in the Stuart Lab is focused on protozoan pathogens and the diseases that they cause. These include malaria which is caused by Plasmodium parasites and Human African Trypanosomiasis (sleeping sickness), Chagas disease and Leishmaniasis that are caused by three Trypanosomatid parasites. The lab investigates novel and essential molecular and cellular processes of the parasites and immune responses to their infections and vaccines to develop drugs, vaccines and diagnostics that are needed.
Malaria Immunology
Dr. Stuart Lab co-leads a multi-institutional U19 research program on human immune responses to malaria, HIV and Covid infection and vaccination and is part of the NIH Human Immunology Project Consortium (HIPC). This project in the lab is focused on systems immunology approaches for the study of human immune responses to Plasmodium falciparum infection and to anti-malaria vaccination. He also co-leads U01 human immunology project that is focused on specific immune cell and immune receptor responses to malaria infection and vaccination and the influence of prior malaria infection. Insights gained from this research program have the potential for impacting strategies for vaccine development and for diagnosing and treating immune-related diseases more broadly.
RNA Editing
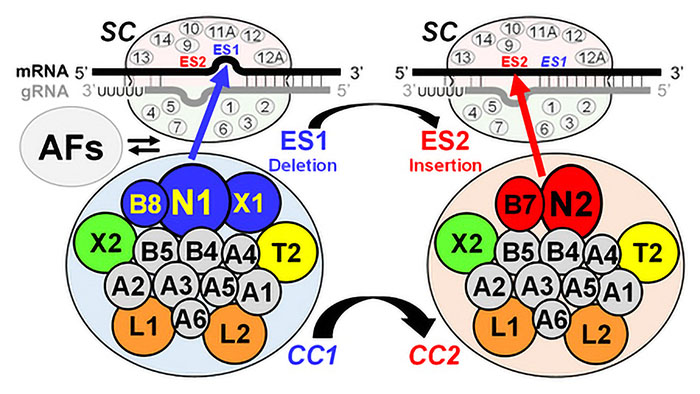
RNA editing is essential to the survival of the three major Trypanosomatid pathogens and does not occur in humans and is thus a source of targets for the development of drugs that are needed to treat infections by these parasites. Elucidating the details of the RNA editing process in the lab has also stimulated discoveries on different types of RNA editing of medical importance, including in humans. The lab determined the many aspects of the editing mechanism and molecular machinery that performs the editing while showing that it is essential in the disease stage and validated several of its components as therapeutic targets. Current studies are characterizing the structure and functions of the proteins and multiprotein complexes that perform editing and are exploring the mysterious mechanisms that result in differential editing in different life cycle stages of the parasite which results in generation of cellular energy by different processes in different stages. Structure/function studies of the proteins and complexes that perform editing will aid the understanding of many nucleic acid processing mechanisms.
Cell Systems
The lab is using a wide variety of cell-wide systems biology approaches to elucidate critical cellular processes in Trypanosomatid parasites that can be exploited for the development of drugs against three related parasites: Trypanosoma brucei, T. cruzi and Leishmania species. It played a leading role in forming the consortium that sequenced and annotated the genomes of these parasitic pathogens; extended this work through high-throughput proteomic and functional genetic studies; identified and validated many drug targets; and created a consortium to develop drugs for these parasitic diseases. The studies include collaborations with medicinal chemists who test numerous compounds for their activity against the drug targets and their potential for development into drugs that may move onto clinical trials.
Antigenic Variation
The lab pioneered research on the process of antigenic variation in trypanosomes, which these parasites use to evade elimination by the immune system, thus preventing the development of a vaccine. The lab discovered that a particular molecular regulatory system controls the selective expression of one of hundreds of surface proteins stage and antigenic switching via epigenetic processes that involves intracellular signaling and chromatin reorganization. It also showed that this regulatory system coordinately controls numerous cellular processes during cell development and that components of the system are conserved from Protozoa through humans within which they have expanded and diverged and control the more complex processes in multicellular organisms including immune responses and functions.
Partnership Opportunities

Kenneth D Stuart, PhD
Ken Stuart, PhD, is a Professor in the Departments of Pediatrics and Global Health in the Schools of Medicine and Public Health at the University of Washington, a P.I. in Seattle Children’s Research Institute's Center of Global Infectious Disease Research and an Affiliate Investigator in the Vaccine and Infectious Disease Division at the Fred Hutchinson Cancer Center. He received a BA in Biology from Northeastern University, a MA in Biology from Wesleyan University and a PhD in Zoology from the University of Iowa. He had Postdoctoral training in Biochemistry at the National Institute for Medical Research, London and at SUNY Stony Brook before becoming an Assistant Professor of Biology at the University of South Florida prior to founding the Seattle Biomedical Research Institute (aka Center for Infectious Disease Research) that merged with Seattle Children’s Research Institute. His expertise is in the molecular and cell biology, immunology and host pathogen interactions with a focus on protozoan pathogens. He is known for his groundbreaking studies of RNA editing, antigenic variation and human immune responses to malaria vaccination and other discoveries (RNA virus). He is also known for his leadership in pathogen genome, drug discovery, and human immunology research consortia. His current research is focused on 1. human immune responses to Malaria vaccination and infection as a leader of a multi-institutional consortium that is also studying responses to HIV, and Covid vaccinations and 2. on in-depth mechanistic studies on RNA editing.
-

Kondalarao Bankapalli, PhD
Fellow
-

Jason Carnes, PhD
Research Scientist IV
-

Senior Research Scientist, Management
-

Jennifer Gilby
Research Technician I
-

Anton Goetz, PhD
Fellow
-

Isaac Lewis, BS
Research Technician I
-

-

-

Paurvi Shinde, PhD
Scientific Project Manager I
-

Varsha Venkatesh
Research Technician I