Smith Lab
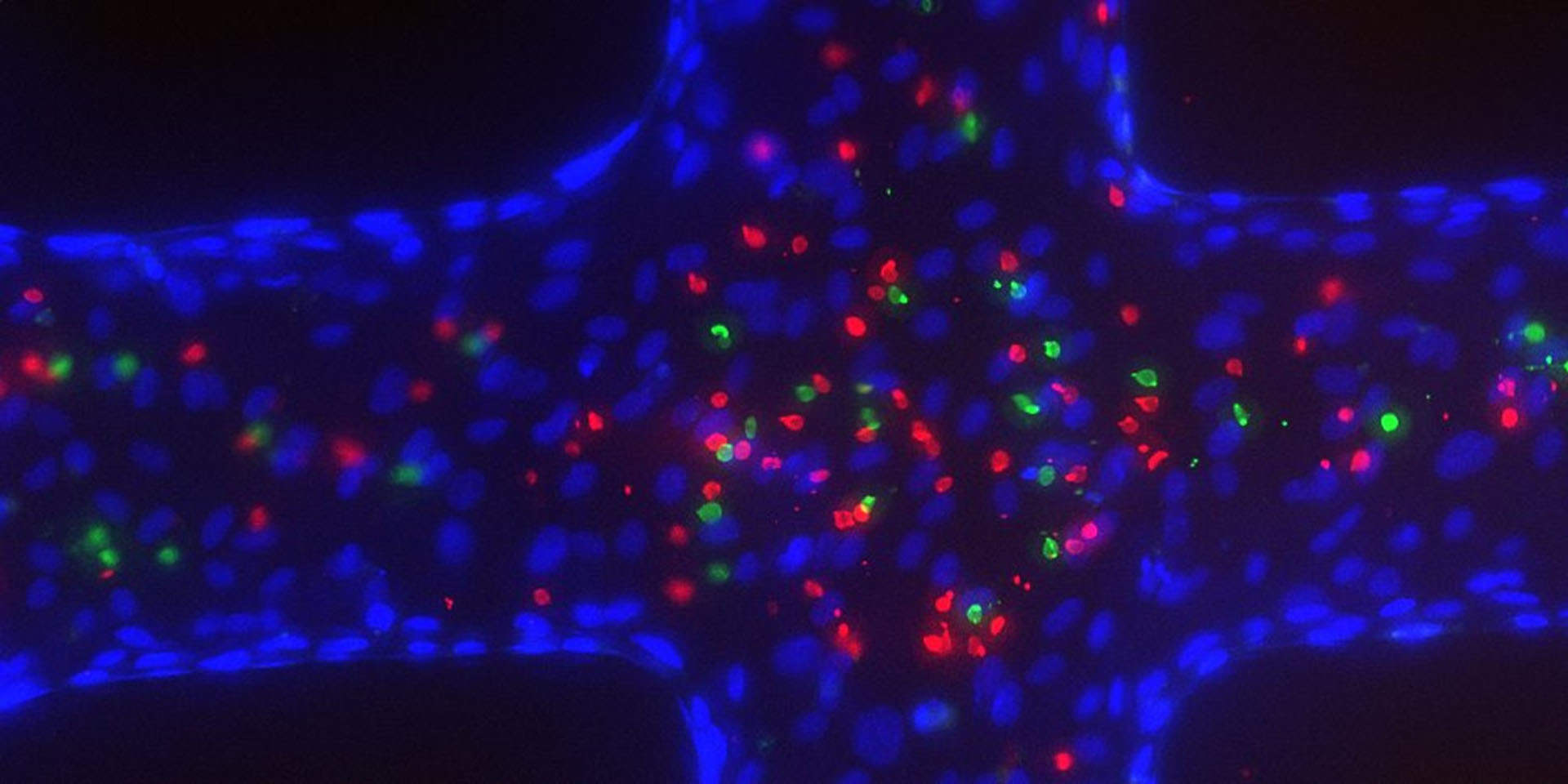
The Smith Lab studies how Plasmodium falciparum malaria parasites cause brain blood vessel damage in cerebral malaria. We use molecular tools and 3D bioengineered brain microvessels to uncover parasite-host interactions and develop new treatments to protect the blood-brain barrier.
Lab News
- September 18, 2025 Priyanka Bansal spoke at the 2025 Molecular Parasitology Meeting
- September 8, 2025 IDSA Daily News Briefing picks featured article by Smith lab postdoctoral fellow
- April 15, 2023 Zheng Lab Unveils Platform for Modeling Cerebral Malaria in 3D - Institute for Stem Cell & Regenerative Medicine
Partnership Opportunities
-
Sara Afereti
Administrative Assistant Senior, Research
-

Priyanka Bansal, PhD
Fellow
-

Luana Dos Santos Ortolan, PhD
Research Scientist III
-

Fatou Joof, PhD
Postdoctoral Scholar
-


