Obado Lab
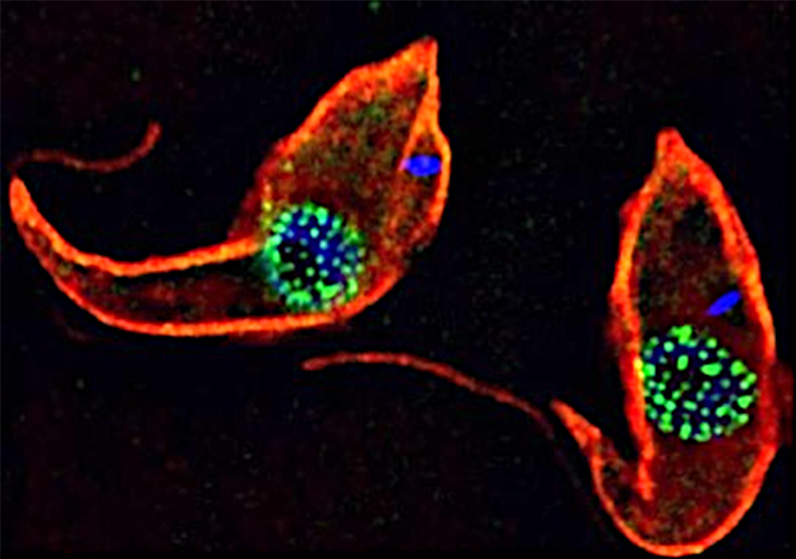
Trypanosomes as models for the study of nuclear pore complex (NPC) structure, function and evolution. Trypanosomes are unicellular flagellated protozoan parasites. Trypanosomes are encased in a subpellicular tubulin array (red – anti tubulin antibody (KMX1)). TbNup89, a nucleoporin component of the NPC has been in situ tagged with GFP to form a TbNup89-GFP chimeric protein (green). DNA is stained blue (DAPI). The DNA outside the nucleus (blue) is the kinetoplast which is the mitochondrial DNA in these organisms. Credit: Jeffrey DeGrasse
The focus of the Obado Lab, led by Sam Obado, PhD, is understanding the functional relationship between the control of gene regulation, RNA export and the nuclear pore complex (NPC), which is the sole mediator of nucleocytoplasmic transport. Among our model systems are trypanosomes, a diverse family of protozoans that are obligatory parasites of invertebrates, vertebrates and plants. Trypanosomes (including Leishmania) cause major public health and economic problems in the developing world and are a growing problem in the southern states of the U.S. in the form of Chagas disease. Trypanosomes are also excavates, early diverging eukaryotes that exhibit non-canonical biological traits, a number of which differentiate them from their vertebrate hosts. Examples include extensive RNA editing, polycistronic transcription and trans-splicing. Thus, studying their cell biology gives deep insights into which systems are conserved, and which are lineage-specific adaptations, across the Eukarya.
The central dogma of molecular biology highlights the flow of genetic information; that DNA is transcribed into RNA in the nucleoplasm and exported through the nuclear pore complex (NPC), to the cytoplasm, where it is translated into protein. However, several components of the transcription-export complex in trypanosomes are cryptic, and the machinery that drives mRNA export in trypanosomes is evolutionarily divergent from those of its vertebrate hosts. Furthermore, the architecture and composition of the trypanosome NPC contains several atypical features, the most unique of which are related to RNA export. Thus, despite being ubiquitous to all eukaryotes, trypanosomes help show us that the NPC has several species-specific subunit arrangements tuned to the biology of each organism. The divergent trypanosome NPC architecture raises questions about the universality of RNA export in eukaryotes and our understanding of RNA export control, which has largely been studied in opisthokonts, such as yeast and vertebrates. This emphasizes Dobzhansky's idea that biology makes sense only in the light of evolution, with trypanosome research shedding light on conserved cell biology principles.
Work in Dr. Obado’s laboratory focuses on uncovering key components of the mRNA export pathway and determining the structure function relationships between nucleocytoplasmic transport and the divergent trypanosome NPC. The goal of the lab is to utilize protein-protein interactome mapping with cell and structural biology to unravel and analyze the composition of the transcription-export complexes and uncover new RNA export pathways in these parasites of public health importance.
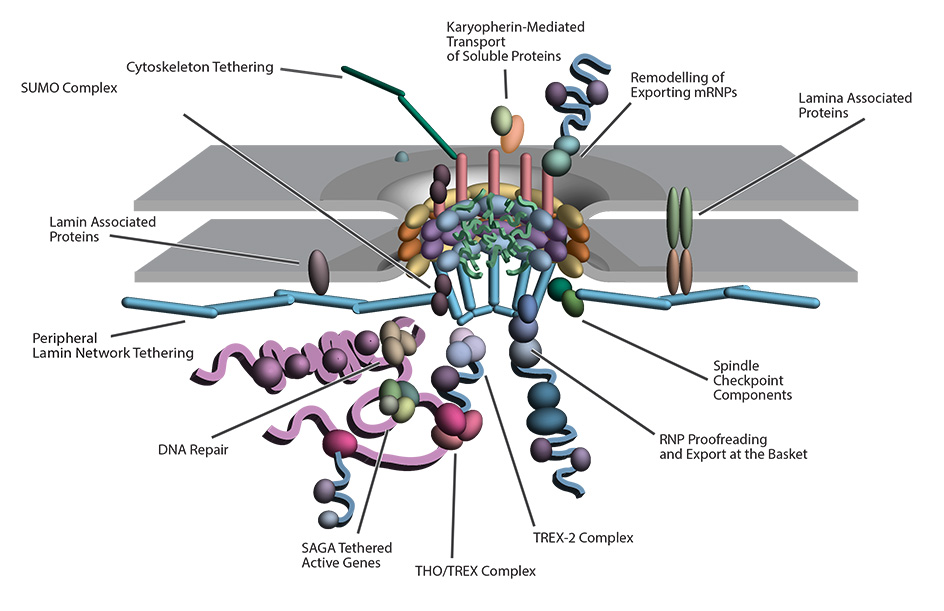
The NPC coordinates several cellular processes through a complex peripheral network. The nuclear pore complex (NPC) associates with numerous molecules and structures in the cytoplasm and nucleoplasm enabling the NPC to be involved in diverse functions in addition to its primary role of import and export of proteins and nucleic acids, which requires nuclear transport factors such as karyopherins (Kaps). The nuclear basket serves as an interaction hub within the nucleoplasm, facilitating various nuclear processes. These include transcriptional regulation, involving interactions between the SAGA chromatin remodeling complex and active genes, as well as interactions between the NPC and components of the TREX2 complex. Additionally, it plays a role in interactions between small ubiquitin-related modifier (SUMO) regulatory complexes and the proofreading machinery of exporting ribonucleoproteins (RNPs), contributes to chromatin stability by engaging with the transcription-coupled DNA repair machinery, and participates in chromosome handling during mitosis through interactions with spindle checkpoint proteins and the spindle. A network of protein–protein interactions extends from the NPC and includes the lamin and nuclear membrane proteins some of which link to cytoplasmic microtubules and coordinate nuclear migration and anchorage, underscoring the extended communication network that spans the nuclear envelope at the NPC and across both the inner and outer nuclear membranes.
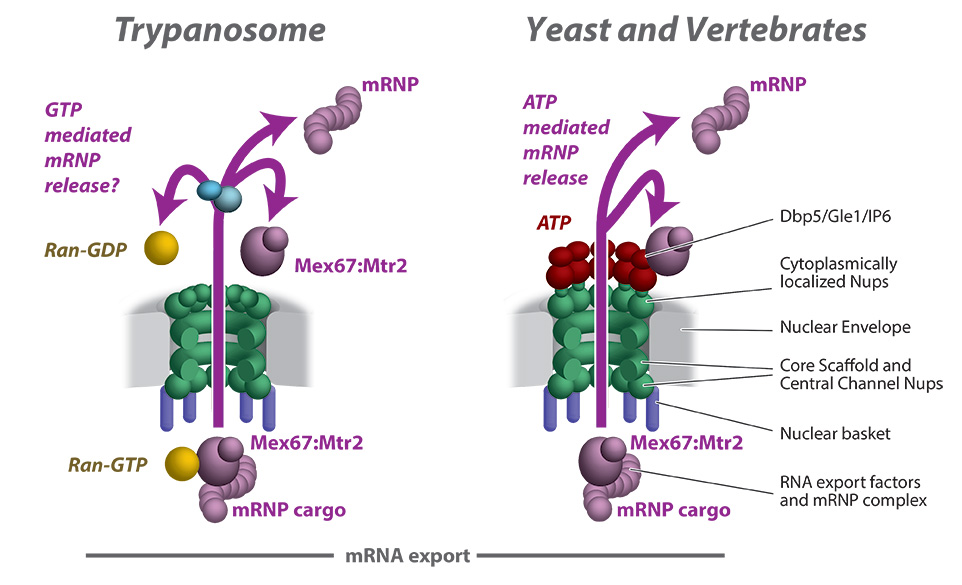
A schematic comparing mRNA export through NPC between trypanosomes and Opisthokonts. The textbook model of mRNA export through the NPC involves the DEAD box helicase Dbp5 and its co-factors Gle1 and inositol hexokinase 6 (IP6) that remodel messenger ribonucleoproteins (mRNPs) and provide directionality to mRNA export. Based on our studies, trypanosomes lack the cytoplasmic Nups that provide an interaction platform for Dbp5 and its cofactors and instead appears to utilize the GTPase Ran to power and provide directionality to mRNA export in trypanosomes. This could be a reflection of their unusual gene transcription which is polycistronic.

